Institute of Experimental and Applied Physics, Christian-Albrechts-Universität zu Kiel
| Metal electroplating: gaining insight into the atomic scale processes by real-time synchrotron x-ray scattering | |
|---|---|
|
|
|
|
Metal electrodeposition processes the formation of metal films by the electrical discharge of ions
from an electrolyte solution play an increasing role in current technologies as well as future applications.
One of many examples is the galvanic deposition of Cu for interconnects on ultra large scale integrated microchips
an important step in the production of modern microelectronic devices. For the fabrication of these microscopic
and nanoscopic metallic structures it is of outstanding importance to understand the complex interplay between
the elemental steps of electrodeposition and the resulting surface morphology. In particular, the influence of
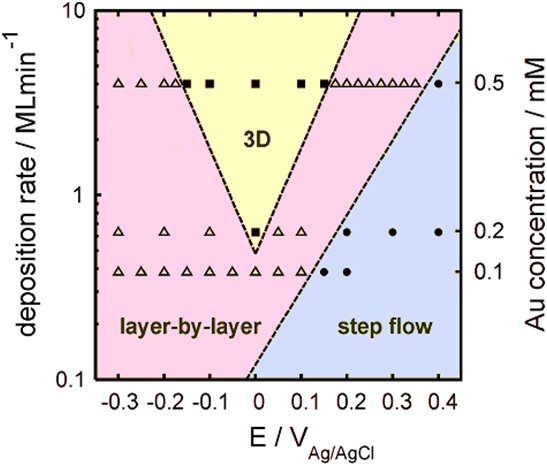
the central deposition parameters such as electrode potential and electrolyte composition is far from clear. We are directly studying how electrochemical deposition progresses on the nanoscale by performing time-resolved surface x-ray scattering (SXS) methods at the interface between metal electrodes and electrolyte solutions during metal growth. Using the high brilliant beams of modern synchrotron sources, such as the ESRF in Grenoble or DESY in Hamburg, as well as custom-build electrochemical cells, designed by our group, and fast detector systems we can simultaneously monitor the x-ray intensity and the electrochemical current with a time resolution in the millisecond range. This permits insight into the type of growth, the roughness evolution at the interface, and the atomic scale interface structure during metal electroplating processes. Currently we aim at realizing experimental conditions which are closer to industrial electrolytic baths for Cu electroplating processes. This includes experiments with comparably high deposition rates and electrolytes containing organic additives. |
|

|
|
| ID32-diffractometer | |
|
|
| growth-diagram | |
Contact: Dr. Jochim Stettner
Funding: DFG project MA 1618/13